|
A compound is a substance composed of two or more elements combined in fixed proportions. When there's only one atom of a given element, we leave off the subscript one. Here we see that an oxygen molecule contains two oxygen atoms, while a water molecule contains two hydrogen atoms and one oxygen atom. A molecular formula is an expression indicating the number of each type of atom in one molecule of a substance. The atoms can be the same, as in the oxygen molecule, or they can be different, as in the water molecule. A molecule is a group of at least two covalently bonded atoms. Instead, most elements exist as molecules or compounds. An ionic bond is a chemical bond formed by the attraction between positive and negative ions.Īs mentioned earlier, atoms rarely exist in isolation in nature, with the except of certain gases like helium or neon. Atoms like sodium and fluorine don't share their electrons. Carbon atoms tend to share electrons with other atoms, such as with four hydrogen atoms in the diagram we see here. A covalent bond is a bond where the electrons are shared between atoms. One type of bond is called a covalent bond. Atoms and ions rarely exist in isolation in nature, because they tend to combine through chemical bonding.Ī chemical bond is an attractive force between two or more atoms or ions that holds them together. Here we see a fluorine atom which accepts an electron to become the negatively charged fluoride ion. This charge means it's a sodium ion.Ĭertain other elements tend to gain electrons. This leaves just 10 electrons, but since there are still 11 protons, sodium has a plus one charge. Sodium is one of several atoms that easily donates an electron. It has 11 protons and 12 neutrons in its nucleus, and 11 electrons outside the nucleus. An ion is a charged species that results from the gain or loss of electrons from a neutral atom or molecule. The most common or stable isotope, such as carbon-12, is frequently known by its element name.Ītoms of some elements readily gain or lose electrons. 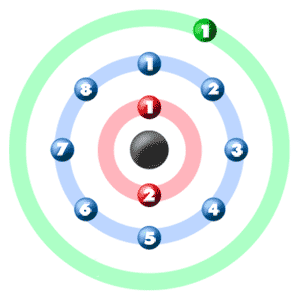
Isotopes are often named according to their atomic mass. The atomic mass is the sum of the number of protons and neutrons, given in atomic mass units or a-m-u’s. The atom on the left has 6 neutrons, the atom in the middle has 7 neutrons, and the atom on the right has 8 neutrons. They all have 6 protons, since they're all carbon atoms.Ītoms are electrically neutral, so they have the same number of negatively charged electrons as positively charged protons. Isotopes are atoms of the same element with different numbers of neutrons in their nuclei. A cloud of negatively charged particles called electrons surrounds the nucleus.Īll atoms of a given element have the same number of protons, but the number of neutrons can vary. The nucleus contains positively charged protons, shown in white, and uncharged neutrons, shown in blue. The nucleus is an atom's center of mass and center of positive charge. An atom is the fundamental unit of chemical matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
